This whitepaper was developed by contextflow’s Scientific and R&D teams to explain our approach behind emphysema quantification and why HU is not the best approach. The full text is listed below. For a pdf copy, click here.
Emphysema quantification with contextflow SEARCH Lung CT
Emphysema detection and volumetry in lung CT serves as an important factor in COPD detection and is relevant for timely, effective treatment and risk prediction for acute respiratory events [González et al. 2018], prognosis [Labaki and Han 2018], or lung cancer surveillance [Sekine et al. 2012]. Recent evidence shows that early COPD detection has clear advantages for patients and is cost effective [Johnson et al. 2021].
Using a threshold of -950 HU is a standard approach for quantification of emphysema in lung CT [Wang et al. 2013]. Although this thresholding method can be used to give an estimate of the emphysema volume, it is not suitable to identify the area of emphysema in detail. By contrast, contextflow SEARCH Lung CT utilizes a deep learning segmentation method to identify the exact area of emphysema and present more accurate volume measurements. When used for detection, the thresholding-based approach identifies emphysema areas in nearly all of the CT scans. In contrast, the AI-based approach has better specificity and thus reduces small false-positive occurrences.
We have assessed the performance of both methods (AI-based Emphysema segmentation by contextflow SEARCH Lung CT; standard HU-based thresholding, no cluster analysis) on 3617 slices drawn from a set of 494 scans with pixel-wise expert annotations. Ground truth was established through manual pixel-wise labeling of emphysematous image regions performed and included a quality control procedure involving expert radiologists.
Results are presented in table 1. The contextflow SEARCH method is more accurate in delineating emphysema locally on a pixel-level. The Dice metric is the standard way to assess the overlap of a segmentation method against the ground truth. The AI method achieves a Dice of 0.4 in contrast to a Dice of 0.23 with the thresholding-based method. Additionally, the volume of emphysema predicted in the slice is closer to the ground truth as can be seen from the volume similarity.

To assess emphysema detection on a case-level, we used a set of 494 positive and 559 negative scans (492 with other pathologies and 67 healthy cases). The contextflow SEARCH method has a similar sensitivity to the thresholding-based approach, detecting nearly every occurrence of the pattern. However the thresholding-based method predicts emphysema in every scan, resulting in 0% specificity. The AI-based method achieves a specificity of 56% and therefore reduces the overhead of false-positive occurrences presented to radiologists.
To better assess the impact of small emphysema predictions, e.g. due to noise in the image, we repeated the evaluation with different coverage cut-offs. Here, emphysema detection is only considered if at least 2%, 5%; or 10% respectively of the overall lung volume is covered with emphysema. Table 2 summarizes the corresponding findings, and shows that the overall difference remains consistent across these cut-offs.

Both datasets include scans with and without contrast enhancement, soft and sharp reconstruction kernels, and a slice thickness ranging from 0.75 to 5 mm. Varying acquisition parameters have been shown to influence the cutoff for emphysema detection [Boedeker et al. 2004, Gierada et al. 2010]; therefore the HU thresholding technique is expected to be more sensitive to the changes in acquisition than the method in contextflow SEARCH Lung CT.
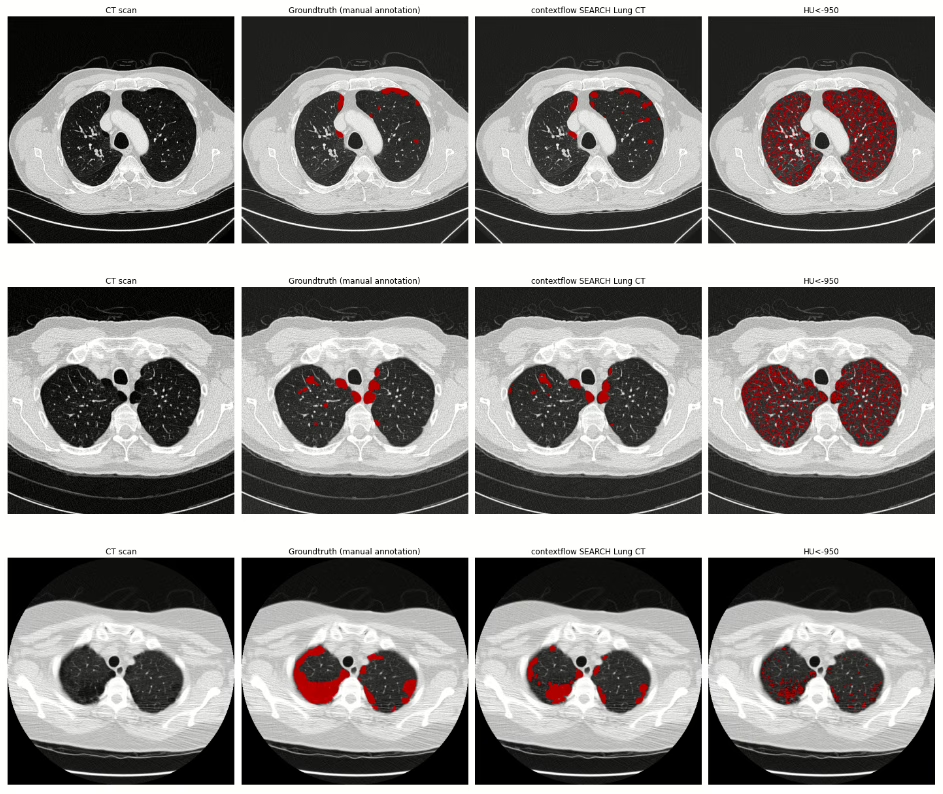
The illustration below provides a visual comparison of results of both approaches for four chest CT scans. The first column (CT scan) shows the image data, the second column visualizes ground truth annotations for emphysema (manual pixel-wise annotations by expert annotators), the third column visualizes segmentation results provided by contextflow SEARCH Lung CT and the fourth column shows segmentation results for the HU-based thresholding approach.


Summary
Estimating the extent of emphysema based on an HU threshold was introduced in the 1990s [Coxson et al. 1990], [Gevenois et al. 1996]. With the recent progress in AI medical imaging, we are now able to measure the extent of emphysema more accurately, which is an important step towards detecting emphysema subtypes on a variety of lung CT scanner types.
In combination with other COPD-relevant patterns [Lynch et al. 2015] that are also supported by contextflow SEARCH Lung CT, this is becoming the new basis for improved decision making in the treatment and progression of COPD patients.
If you have any questions, comments, are interested in a partnership or getting contextflow integrated into your clinical routine, feel free to contact us at office@contextflow.com.
References
Boedeker KL, McNitt-Gray MF, Rogers SR, Truong DA, Brown MS, Gjertson DW, Goldin JG. Emphysema: effect of reconstruction algorithm on CT imaging measures. Radiology. 2004 Jul;232(1):295-301.
Gierada DS, Bierhals AJ, Choong CK, Bartel ST, Ritter JH, Das NA, Hong C, Pilgram TK, Bae KT, Whiting BR, Woods JC. Effects of CT section thickness and reconstruction kernel on emphysema quantification: relationship to the magnitude of the CT emphysema index. Academic radiology. 2010 Feb 1;17(2):146-56.
Gevenois PA, De Vuyst P, de Maertelaer V, et al. Comparison of computed density and microscopic morphometry in pulmonary emphysema. Am J Respir Crit Care Med. 1996;154(1):187–192. doi:10.1164/ajrccm.154.1.8680679
Coxson HO, Rogers RM, Whittall KP, et al. A quantification of the lung surface area in emphysema using computed tomography. Am J Respir Crit Care Med. 1999;159(3):851–856. doi:10.1164/ajrccm.159.3.9805067
Sekine, Y., Katsura, H., Koh, E., Hiroshima, K. and Fujisawa, T., 2012. Early detection of COPD is important for lung cancer surveillance. European Respiratory Journal, 39(5), pp.1230-1240.
Lynch DA, Austin JH, Hogg JC, et al. CT-definable subtypes of chronic obstructive pulmonary disease: a statement of the Fleischner Society. Radiology. 2015;277(1):192–205. doi:10.1148/radiol.2015141579
Labaki, W.W. and Han, M.K., 2018. Improving detection of early chronic obstructive pulmonary disease. Annals of the American Thoracic Society, 15(Supplement 4), pp.S243-S248
González, G., Ash, S.Y., Vegas-Sánchez-Ferrero, G., Onieva Onieva, J., Rahaghi, F.N., Ross, J.C., Diaz, A., San José Estépar, R. and Washko, G.R., 2018. Disease staging and prognosis in smokers using deep learning in chest computed tomography. American journal of respiratory and critical care medicine, 197(2), pp.193-203.
Johnson, K.M., Sadatsafavi, M., Adibi, A., Lynd, L., Harrison, M., Tavakoli, H., Sin, D.D. and Bryan, S., 2021. Cost effectiveness of case detection strategies for the early detection of COPD. Applied Health Economics and Health Policy, 19(2), pp.203-215.